Your lab’s workflow accelerates because the process is automated and transparent, and you can see business results sooner because the implementation process for SLIMS is rapid.
A LIMS Made for Biobanks
Agilent SLIMS provides a comprehensive software that manages
and tracks all biobank samples over their entire lifetime.
Learn more by watching this video

Collect. Store. Discover.
Agilent SLIMS for Biobanks brings a consistent, holistic approach to managing your lab’s biospecimen samples, improving the quality of research and the reliability of results. SLIMS provides a fast, easy way to manage your biological materials, and to implement quality control measures to be taken during sample collection, prior to sample accession, and upon sample use, ensuring the highest quality possible for your biobank.
Get startedSLIMS advantages for Biobanks
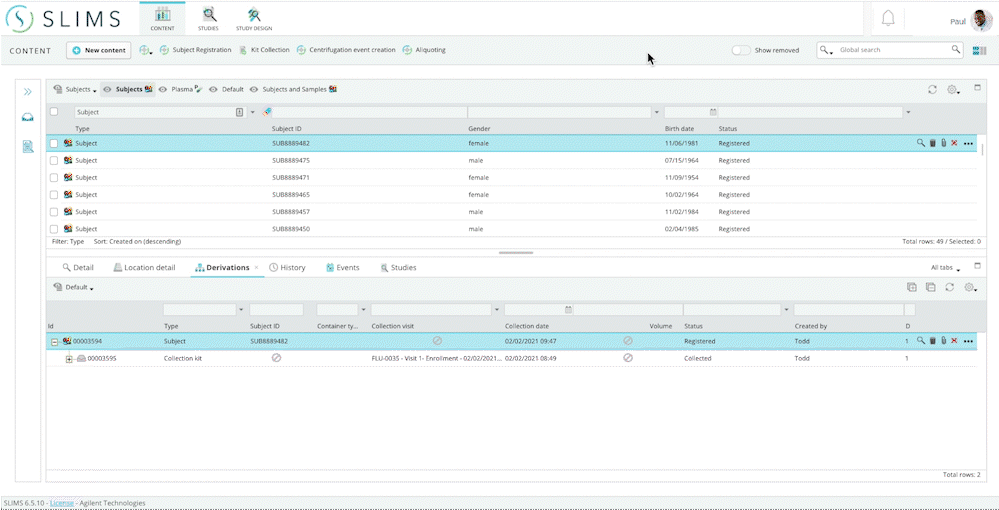
SLIMS makes it easy to create studies, register subjects, schedule visits for sample and kit collections, create events on the specimens, filter and highlight important sample data. All sample information can be managed, such as sample type, its container, location, statuses, or any other important details. You can also monitor all events and changes that happened over time, including when the change was made and by whom. SLIMS makes data instantly accessible to authorized lab personnel and managers, even remotely.
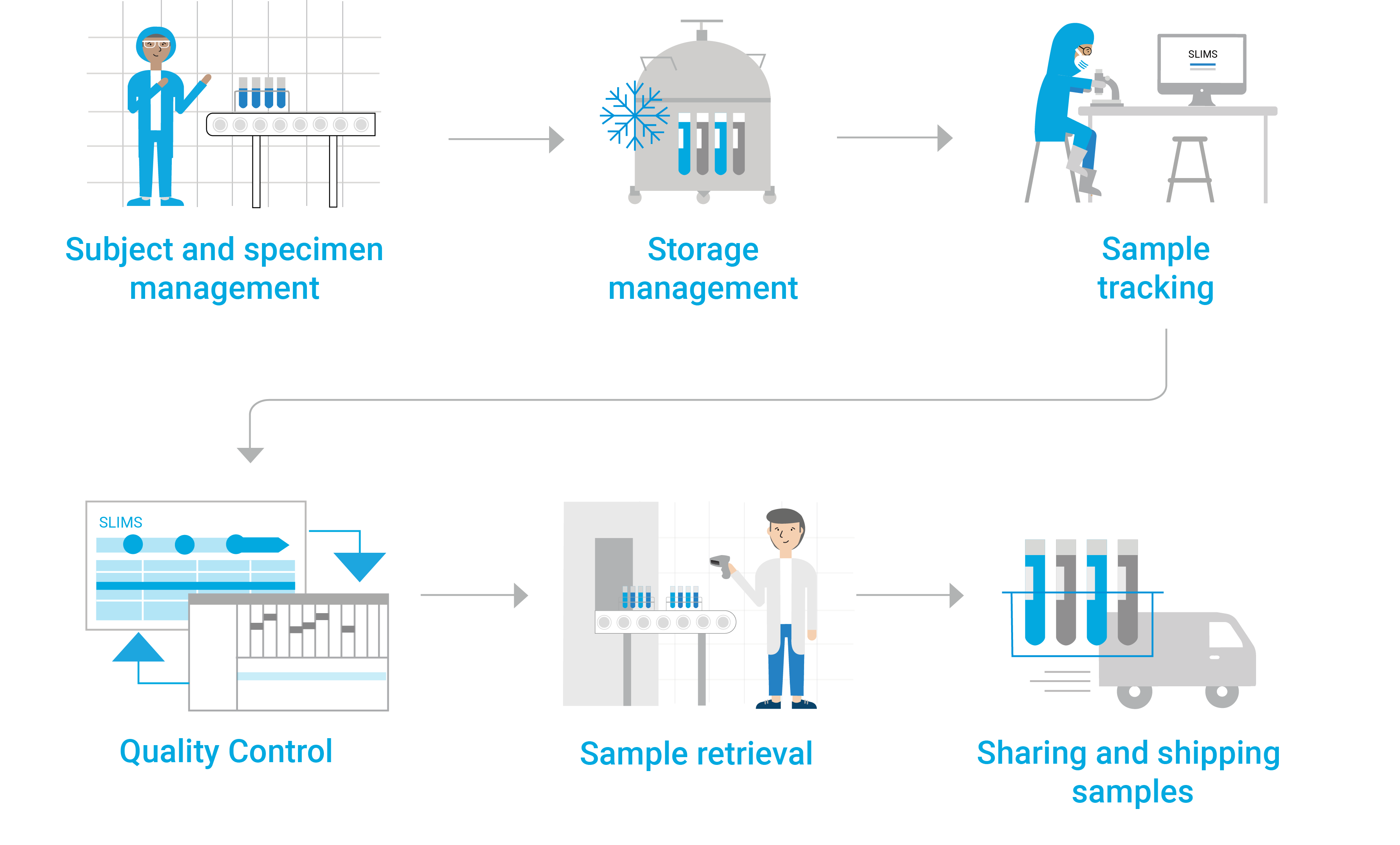
Biobank Sample Life Cycle
With SLIMS, you can integrate instruments in your lab, such as TapeStation, and connect with other Agilent and non-Agilent lab software systems. Results from these systems are easily transferred to SLIMS, and the data can be reused immediately.
This makes it easier to track and manage lab resources, and SLIMS monitors access to these resources based on your own rules. In short, you always have an accurate overview of the live status of your lab. And you can easily restrict usage and grant authorization to ensure that the right instruments and tools are being used.
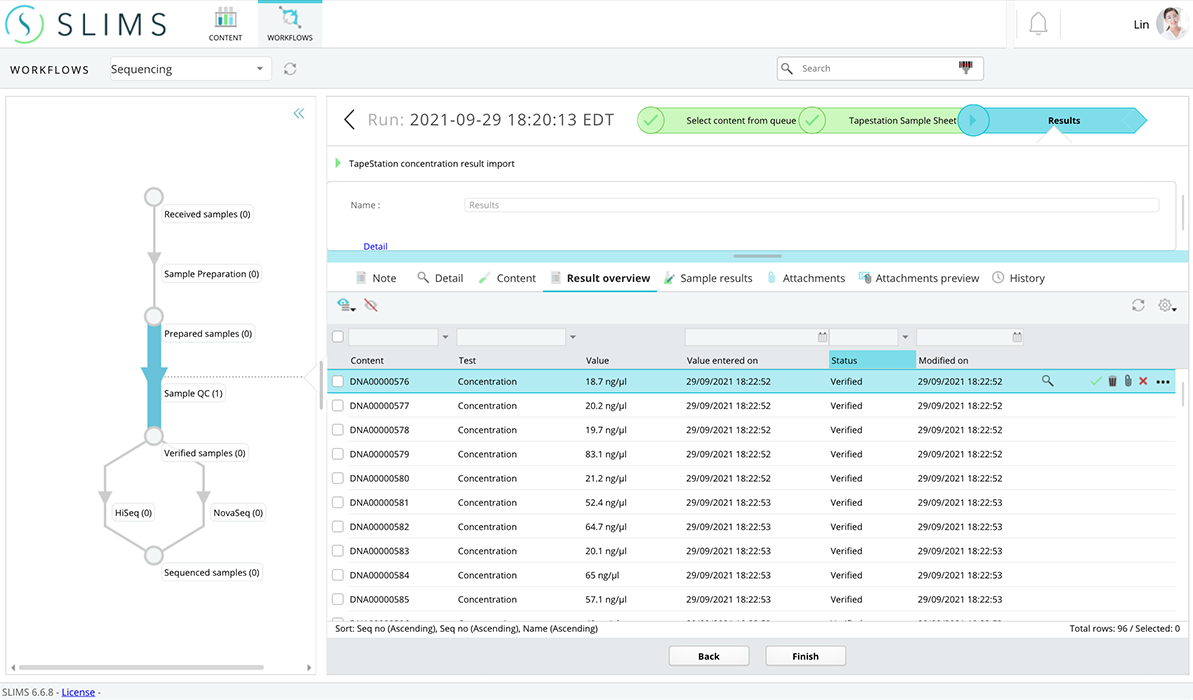
Connection with Agilent TapeStation software is automatic and results are conveyed to SLIMS.
SLIMS can support ISO 20387:2018 requirements, as well as stringent or legally required standards as dictated by specific markets. SLIMS also supports ISBER guidelines for quality control (QC) across all repositories and specimen types, and Standard PREanalytical Code (SPREC), corresponding to the most critical preanalytical variables of fluid and solid biospecimens. QC processes include the four “pillars” of collection quality:
- Authenticity—correctly assigned identity
- Purity—freedom from contamination
- Stability—capability of sample material to retain the initial value of a measured quantity for a defined period of time within specific limits when stored under defined conditions
- Consent—for human specimens
What our clients say
Hear what our clients have to say about their experience with SLIMS

“SLIMS helps the fly facility track orders from both internal and external clients. Using SLIMS, users who are looking for a specific strain of fruit fly can browse the collection available through the FlyORF facility, select their desired strain, and place an order all in the same system. The multifaceted ability of SLIMS makes this possible from the front end while also controlling all the ordering, sample management, and billing for the lab on the back end. It also saved the FlyORF lab money and time by reducing the need to purchase multiple systems.”

“Agilent SLIMS was selected to manage our biobank and cohort, establishing the continuous observation and follow-up of the chosen subject pool. SLIMS’ features makes it easy to enroll subjects and monitor their progression in the cohort and to provide easy access across different institutions, yet comply with data protection and subject protection needs.”
Frequently Asked Questions
Biobanks are documented collections of biological material used for research. Also called tissue banks, biorepositories, or biospecimen repositories, biobanks exist for a broad range of sample types from plant, animal, environmental, and many other sources. Biobanks typically involved in medical research store samples of human origin, such as body fluids, tissue samples, and even whole organs, that are collected through routine or other medical procedures.
In addition to providing access to human samples and their associated records, biobanks may offer access to a range of medical information, including genomic data, population-genetic data, and molecular data. Access to this kind of information is critical to many medical research laboratories that may not have the resources to maintain such repositories.
Medical research biobanks contain human samples from large numbers of people, and they are used to explain how genes and the environment interact in order to prevent diseases or treat them more effectively. The genetic information from each sample can be linked to the individual’s medical history and other personal factors that impact health (e.g., diet, smoking etc.). In these biobanks, human samples are normally saved for many years.
Human biospecimens are any biological material taken from a human for diagnostic or research purposes. Biospecimens that are retained for research include blood and other body fluids (plasma, serum, urine, feces, sputum, bile, cerebrospinal fluid (CSF), etc.), solid samples of tissues and organs, as well as gametes, embryos, and fetal cells. These samples may be collected from healthy people as well as those with disease conditions during clinic visits, surgical procedures, and autopsies.
Biospecimens are useful for connecting research findings to patient data, enabling the search for better diagnostic and treatment strategies. Human biospecimens are a great aid for medical researchers looking to:
- Determine who may be likely to develop certain diseases
- Understand diseases progress and variation
- Determine a patient's likelihood to respond to specific medicines
- Investigate appropriate treatments
- Improve screening tools for detection and treatment
Biological samples contain a host of information that is critical for understanding diseases and their treatments. DNA, glycans, proteins, disease biomarkers, and other molecules of interest can all be assayed, and the results can be directly connected to detailed patient data, providing insight that is often unavailable through other approaches.
Discovering and developing new ways to diagnose and treat disease are central to medical progress. Samples that are collected, documented, and stored under strictly controlled conditions are a vital part of this process.
Medical research on human samples contributes to many important outcomes, including:
- Understanding how and why patient characteristics correlate with responses to a given treatment and establishing characteristic profiles of patients who should not receive certain drugs can improve diagnosis and therapy.
- Establishing molecular biomarkers to serve as indicators that can be detected in human samples can help improve monitoring disease and treatment progress; discovering molecular indicators of disease progression can also help identify new drug targets.
- Understanding disease origins, including genetic links, can improve our ability to avoid or delay disease onset.
The activity of medical biobanks is strictly controlled by regulatory guidelines created to ensure quality and secure operations. Biobanks collect, store, and process samples and their associated data, and make these available for approved medical research projects that have demonstrated appropriate scientific intent and regulatory compliance.
Different types of biobanks, such as those integrated into hospitals and population-based biobanks, are available to serve the needs of different areas of medical research. Both serve the goal of improve healthcare. Population-based biobanks house samples collected from healthy volunteers from the general population, and routinely follow up by continuing to collect data on the donors over decades. This can be particularly helpful in studies of predisposing factors or susceptibilities.
In contrast, donors to hospital-integrated biobanks collect already have (or are suspected of having) a disease. Research groups can access these medical archives, thus avoiding the time and expense involved in collecting and storing the samples themselves.
Standards defined under ISO 20387:2018 outline the general requirements for the competence, impartiality, and consistent operation of biobanks, including those needed to ensure the quality of biological material and associated data. Effective practices for the management of biological and environmental specimen collections and repositories are detailed in International Society for Biological and Environmental Repositories (ISBER) Best Practices: Recommendations for Repositories (4th Ed.).
European institutions also have well-defined guidelines to regulate biobanking practice, including laws related to data privacy. Best practice guidelines have also been released by OECD, International Agency for Research on Cancer (IARC), and other WHO agencies.
However, it is important to note that biobanking in the European Union (EU) currently falls under other existing regulatory frameworks rather than specific biobanking laws. Substantial differences in laws between countries can complicate the exchange of human samples between European countries and may impede international medical research collaborations. hSERN is a valuable resource for specific information on legislation in place in countries across the EU.
All these requirements impact the procedures needed to handle the samples and record all events involving those samples, as well as on the procedures needed to assess sample quality. Software solutions specifically designed to support these requirements are a necessity for biobanks that cannot rely on standard data management systems.
To ensure the safety and quality of samples and their associated personal data, storage must fully comply with all regulations relevant to the storage location. Access is only available to authorized staff. Samples/data requested for medical research can be accessed only after the requesting scientist has received permission from the appropriate Internal Review Board (IRB), Medical Ethical Committee (MEC), or local equivalents.
Software solutions can assist with data access management so that only authorized persons see only what they need to know. For example, a technician might not require access to the personal history of the patient from whom the sample originated, but might need to know:
- Potential presence of pathogens or communicable disease agents, in order to ensure appropriate precautions are taken during subsequent handling and analysis.
- The conditions the sample has been exposed to, in order to ensure sample quality meets the requirements for analyses.
For research performed within a large multicenter research project, samples may be shipped to other institutes. Local regulations in these destinations may differ from those of the originating biobank; they may require that samples be returned to the biobank, but it is also possible that any leftover material may be destroyed, upon prior explicit agreement.
When samples are provided for research, the information related to them must be
coded. Samples not used for the
patient's treatment or future diagnosis are provided anonymously, with no
possibility to link the sample to the
patient's personal data.
Additionally, biobanks also implement measures to mitigate incidents such as fire or
flooding that could impact the
samples and provide measures to track and assess the quality of sample storage
conditions.
Patients may have certain rights related to their samples. For example, they may ask
to be kept informed about how
their sample and data will be used, as well as other questions of storage duration,
security, and access. Patients
may also provide information relating to their health, nutrition, and lifestyle or
allow that information to be obtained
from their medical records.
Biobanks use software to collect and manage patient personal/private information;
such software must conform to
guidelines as set forth by relevant governing bodies, such as Health Insurance
Portability and Accountability Act
(HIPAA) in US, General Data Protection Regulation (GDPR) in the EU, Act on the
Protection of Personal Information
(APPI) in Japan or Personal Data Protection Commission in Singapore.
Because of the sensitive nature of the data, software must follow strict security norms such as ISO/IEC 27001. Automated and secure connection between the biobank storage solution and the in-place Electronic Medical Record (EMR) solution in the originating hospital provide benefits for productivity and quality (e.g., no need to re-enter data, fewer copy-and-paste errors).
The main risks for participants in biobanking surround privacy, the potential for stigmatization and discrimination, and dealing with incidental findings.
The fact that clinical patient data as well as the samples themselves may be needed to perform medical research impacts issues of patient privacy. While it is not always essential that the identity of the patient be known, being able to link the sample and data to the patient and treating doctor may be required in certain instances. In addition, some mechanism for collecting follow-up data related to the patient's experience, clinical response, and treatment outcome may be required. In these instances, coding is employed to protect personal data while providing unrestricted clinical examination.
Stigmatization and discrimination are also potential concerns with biobanking, as samples contain genetic material that can reveal information such as disease predisposition.
Biobanks may be able to provide feedback to donors in the case of relevant research findings; however, practices vary, so patients may opt to inquire about the policy of the biobank to which they might contribute. Incidental findings are unexpected results that dispute or change the original diagnosis and, potentially, the chosen treatment options. Practices also vary on if and how this information is communicated back to the patient and their doctor. Any reported findings are submitted through the treating doctor, who can evaluate how these findings might impact the original diagnosis. Incidental findings should be evaluated in the context of the reliability of the performed test; questionable results may require repeat testing using new material.
All personal data related to a sample donor (e.g., name, address) is highly regulated and regarded as strictly confidential. Access is strictly limited, and sample information is coded to protect personal identifiers under layers of encryption. The coding enables re-identification in the case of findings that need to be communicated to the treating physician, and for associating any relevant additional data.
As the laws that govern personal data protection vary between nations, specific enquiries about data security should be directed to the biobank.
A patient who consents that their samples can be saved may change their decision through a ‘withdrawal of consent’ or ‘opt-out’. Consent may be withdrawn at any time; the patient's doctor and the designated contact at the biobank must be notified, as appropriate.
Local laws differ in how this procedure is handled. Some may require the biobank to offer the option to remove samples and related information from the biobank, or that the biobank may retain them in a completely nonidentifiable form. Samples are unlikely to be returned to the patient even if requested. Likewise, results from samples that have already been used for medical research usually cannot be retracted from the biobank.
Biobanks play a crucial role in medical research by having robust and even rare biospecimen samples readily available for clinical studies. As sample collection activity and the number of sources increase, and storage requirements (e.g., tenure, security, regulatory scrutiny) become more complex, the need for implementing a digital solution to manage the biobanking process with rigor and consistency is obvious.
With SLIMS, Agilent provides a comprehensive software solution that manages and tracks all biobank samples over their entire lifetime. This includes quality control measures to be taken during sample collection, prior to sample accession, and upon use of the sample, to ensure biological material and data collections of the highest quality possible. SLIMS also provides the sophisticated controls needed to ensure data remain private.
Getting a demo is easy.
Fill out the form and one of our experts will contact you.